Density is a measure of mass per volume represented by this formula! The units are interchangeable, so if you need to figure out mass or volume, you can rearrange and solve!
Density is measured in g/l or g/ml.
Today we also learned about the density of Gases. They vary with temperature. The formula for finding the density of a gas is mm(molar mass)/ stp(22.4l)
Chemistry 11 Mr. Doktor Block G Run by: Mark W. Guarin Julius O. Francia Timothy Samson
Monday, December 13, 2010
Atoms-Molecules-Moles
Moles
^
Molecules(chemical formula of an element)
^
Atoms(what matter is made up of)
Awhile back, we learned how to go from atoms to molecules to moles. It's hard to explain, so let's do an example.
How many atoms are there in 1.5mol of Iron?
So first off, we write down what we know.
1.5Mol
You want to cancel moles, so you put moles on the bottom, and since you want atoms, you put atoms on top. Remember 1atom is equal to avogadro's number which is 6.02x10^23.
1.5mol x 6.02x10^23/1mol which equals 9.03x10^23 atoms, remember significant digits! = 9.0x10^23 atoms!
What's the difference between atoms and Molecules?
Molecules are composed of atoms, for example Lithium's atomic mass is 6.9.If an element is diatomic such as Carbon, it is 12x2.
(HOFBRCl)
^
Molecules(chemical formula of an element)
^
Atoms(what matter is made up of)
Awhile back, we learned how to go from atoms to molecules to moles. It's hard to explain, so let's do an example.
How many atoms are there in 1.5mol of Iron?
So first off, we write down what we know.
1.5Mol
You want to cancel moles, so you put moles on the bottom, and since you want atoms, you put atoms on top. Remember 1atom is equal to avogadro's number which is 6.02x10^23.
1.5mol x 6.02x10^23/1mol which equals 9.03x10^23 atoms, remember significant digits! = 9.0x10^23 atoms!
What's the difference between atoms and Molecules?
Molecules are composed of atoms, for example Lithium's atomic mass is 6.9.If an element is diatomic such as Carbon, it is 12x2.
(HOFBRCl)
Tuesday, November 23, 2010
Moles And Volume of a Gas
Mass <---> Moles <---> Volume
Ex. How many liters will 5.0 mol occupy at STP.
5.0Mol * 22.4L
-------- = 112 L
1 Mol
Ex. At STP a sample of Nitrogen gas contains 15.5. How many liters are there?
15.5 Mol * 22.4L
-------- = 347.2 L
1 Mol
Ex. STP an Hydrogen gas occupies 12 liters. How many moles of gas must be there?
12L * 1 Mol
-------- = 0.54 Mol
22.4L
- At a specific pressure and temperature one mole of gas occupies the same volume
- At 0'C 101.3 KPa 1 Mole = 22.4L
- This temperature and pressure is called STP
- 22.4L/Mol is the molar volume at STP
Ex. How many liters will 5.0 mol occupy at STP.
5.0Mol * 22.4L
-------- = 112 L
1 Mol
Ex. At STP a sample of Nitrogen gas contains 15.5. How many liters are there?
15.5 Mol * 22.4L
-------- = 347.2 L
1 Mol
Ex. STP an Hydrogen gas occupies 12 liters. How many moles of gas must be there?
12L * 1 Mol
-------- = 0.54 Mol
22.4L
Monday, November 22, 2010
Molar Mass
Molar Mass - The mass of one mole of a substance. (Mass is in grams)
- Measured in g/mole
- Mass could be found using the atomic mass on the periodic table
Molar Mass of Compounds
To find the molar mass of compounds add the mass of all atoms.
Element Molar Mass
H20 2(1.0) + 16.0 = 18.0 g/mol
NO2 14.0 + 2(16.0) = 46.0 g/mol
NaCl 23.0 + 35.5 = 50.5 g/mol
*Always remember Significant Digits.
Then there is converting between grams and moles
We use molar mass as the conversion factor to convert mass and mole.
Example
Find the mass of 0.89mol of CaCl2
0.89mol x 111.1 g = 98.879g = 99g 40.1 + 2(35.5) = 111.1
------------
1 mol
Determine the number of moles of C5H12 that are in a 362.8g of the compound.
362.8g x 1 mol = 5.039mol 5(12.0) + 12(1.0) = 72.0
--------------
72.0 g
- Measured in g/mole
- Mass could be found using the atomic mass on the periodic table
Molar Mass of Compounds
To find the molar mass of compounds add the mass of all atoms.
Element Molar Mass
H20 2(1.0) + 16.0 = 18.0 g/mol
NO2 14.0 + 2(16.0) = 46.0 g/mol
NaCl 23.0 + 35.5 = 50.5 g/mol
*Always remember Significant Digits.
Then there is converting between grams and moles
We use molar mass as the conversion factor to convert mass and mole.
Example
Find the mass of 0.89mol of CaCl2
0.89mol x 111.1 g = 98.879g = 99g 40.1 + 2(35.5) = 111.1
------------
1 mol
Determine the number of moles of C5H12 that are in a 362.8g of the compound.
362.8g x 1 mol = 5.039mol 5(12.0) + 12(1.0) = 72.0
--------------
72.0 g
Wednesday, November 17, 2010
Avogadro's number (how we count atoms)
Atoms and molecules are really really small, so it would take a very very long time to count them.
How do we measure them then?
A man named Amedeo Avogadro proposed that the number of atoms in 12 g's of carbon can be equal to a constant(which is equal to 1 mol of carbon).
What is a mol? No not a mole!
A mole(mol) is just a unit of measurement for atoms, molecules and formula units, like how a dozen is equal to twelve or how a millennium is 1000.
What is Avogadro's #?
 So how big is a mol? If we used real life a real life example, 1 mol of meters would cross the galaxy over 3000 times.
So how big is a mol? If we used real life a real life example, 1 mol of meters would cross the galaxy over 3000 times.
How do we measure them then?
A man named Amedeo Avogadro proposed that the number of atoms in 12 g's of carbon can be equal to a constant(which is equal to 1 mol of carbon).
What is a mol? No not a mole!
A mole(mol) is just a unit of measurement for atoms, molecules and formula units, like how a dozen is equal to twelve or how a millennium is 1000.
What is Avogadro's #?
 So how big is a mol? If we used real life a real life example, 1 mol of meters would cross the galaxy over 3000 times.
So how big is a mol? If we used real life a real life example, 1 mol of meters would cross the galaxy over 3000 times. Example of figuring out how many moles is in an element
A sample of Hydrogen contains 2.2 times 10 to the power of 21 atoms. How many moles of carbon is this?
In order to figure out this problem, we use dimensional analysis. You want to figure how many moles there are and so to do this, you want to cancel out atoms, so you use Avogadro's number!
So you write 2.2 x 10^21 atoms times 6.02 x 10^23 moles/atoms. The atoms cancel each other out and you end up with 1.32 x 10 to the power of 45!
Monday, November 8, 2010
Second Lab Hydrate Lab
Hydrates are ionic compounds that contain an inorganic salt compound loosely bound to water.
The purpose of this lab was to determine the practical formula of a hydrate.
In this lab we determined the mass of the hydrate without water (anhydrous)
We used:
-Bunsen Burner
-Test tube
-Test tube clamp
-Test tube rack
-Weight scale
-Cobaltous Chloride Hexahydrate
First we filled the test tube with Cobaltous Chloride Hexahydrate then weighed it.
Then we began to use the Bunsen burner while using the clamps to hold the test tube over it.
We noticed the Cobaltous Chloride Hexahydrate turn from the color red to a light blue.
Then we re-weighed the test tube.
The percent of water in Cobaltous Chloride Hexahydrate was 45%.
-chloride-hexahydrate-sample.jpg/220px-Cobalt(II)-chloride-hexahydrate-sample.jpg)

_chloride.jpg)
The purpose of this lab was to determine the practical formula of a hydrate.
In this lab we determined the mass of the hydrate without water (anhydrous)
We used:
-Bunsen Burner
-Test tube
-Test tube clamp
-Test tube rack
-Weight scale
-Cobaltous Chloride Hexahydrate
First we filled the test tube with Cobaltous Chloride Hexahydrate then weighed it.
Then we began to use the Bunsen burner while using the clamps to hold the test tube over it.
We noticed the Cobaltous Chloride Hexahydrate turn from the color red to a light blue.
Then we re-weighed the test tube.
The percent of water in Cobaltous Chloride Hexahydrate was 45%.
-chloride-hexahydrate-sample.jpg/220px-Cobalt(II)-chloride-hexahydrate-sample.jpg)

_chloride.jpg)
Thursday, November 4, 2010
Chemical Nomenclative
Chemical Formulas
- Be aware of the difference between ion and compound formulas
- Same elements can form more than one ion
- the top number on the periodic table is more common
- IUPAC uses roman numerals in parenthesis to show the charge
- Classical system uses Latin names if the element and the suffixes ic (Larger charge) and ous (Smaller charge)
- Ferr - Iron
- Cupp - Copper
- Mercur - Mercury
- Stann - Tin
- Aunn - Gold
- Plumb - Lead
- Complex ions are larger groups of atoms that stay together during a chemical reaction
- Almost all are anions (Negative charge)
- Some compounds can form lattices that bond to water molecules
- These crystals contain water inside them which can be released by heating
- To name hydrates
- Write the name of the chemical formulas
- Add a prefix indicating the number of water molecules (mono=1, di=2, tri=3)
- Add the hydrate after prefrix
- Hydrogen compounds are acid
- Hydrogen appears first in the formula unless it is part of polyatomic group
- If there is OH in the formula it is a base
Electronic Structure
Drawing Electron dot diagrams:
The nucleus is represented by the atomic symbol for individual elements of valence electrons.
Electrons are represented by dots around the symbol.
There are four orbitals each holding a maximum of electrons. Each orbital gets at least 1 electron before they pair up.
Lewis Diagrams for compounds & Ions
To do a lewis diagram for compounds & ions
1) Determine the # of valence electrons for each atom in the molecule
2) Place atoms so that valence electrons are shared to fill each orbital
The nucleus is represented by the atomic symbol for individual elements of valence electrons.
Electrons are represented by dots around the symbol.
There are four orbitals each holding a maximum of electrons. Each orbital gets at least 1 electron before they pair up.
Lewis Diagrams for compounds & Ions
To do a lewis diagram for compounds & ions
1) Determine the # of valence electrons for each atom in the molecule
2) Place atoms so that valence electrons are shared to fill each orbital
Hydrogen has an extra one and chlorine needs one.
They form a covalent bond and share electrons.
Double and triple bonds:
Sometimes the only way covalent compounds can fill all their valence electron levels is if they share more than 1 electron.
Sunday, October 31, 2010
What exactly is on the Periodic Table?
On a periodic table, elements that are close to each other on the table display similar characteristics.
There are 7 important periodic trends:
1. Reactivity
2. Ion charge
3. Melting Point
4. Atomic Radius
5. Ionization Energy
6. Electro Negativity
7. Density

Reactivity
Non-metals and metals show different characteristics
The most reactive metal is Francium
The most reactive non-metal Fluorine

Ion Charge
Ion Charges of elements depend on their group (columns).
+1|+2 <-----The Variable Changes----->+3|+/-4|-3|-2|-1|0

Melting Point
The elements in the middle of the periodic table have the highest melting point.
Noble Gases have the lowest melting point.
Starting from the <- and moving to the ->, the melting point increases until it gets to the middle of the table.

Atomic Radius
The radius decreases when moving on the table, up and to the right.
Francium has the largest atomic radius.
Helium has the smallest atomic radius.
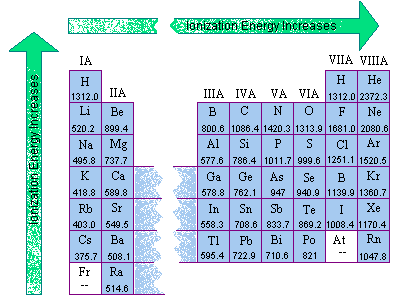
Ionization Energy
Ionization Energy is the energy needed to completely remove an electron from an atom.
Ionization Energy increases going up and the right on the table.
All the noble gases have a high ionization energy.
Francium has the lowest Ionization Energy.
Helium has the highest Ionization Energy.
It's the opposite trend from atomic radius.
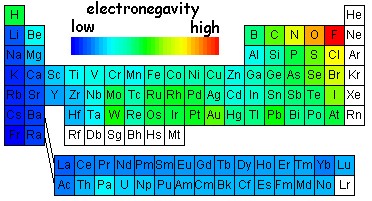
Electronegativity
Electronegativity refers to how much atoms want to electrons.
It has the same trend as Ionization Energy.

Density
A measure of the quantity of some physical property (usually mass)per unit length, area, or volume (usually volume).
Energy density is a measure of the amount of energy (often inthe form of electromagnetic radiation) per unit volume in a region of space or some material.
There are 7 important periodic trends:
1. Reactivity
2. Ion charge
3. Melting Point
4. Atomic Radius
5. Ionization Energy
6. Electro Negativity
7. Density

Reactivity
Non-metals and metals show different characteristics
The most reactive metal is Francium
The most reactive non-metal Fluorine

Ion Charge
Ion Charges of elements depend on their group (columns).
+1|+2 <-----The Variable Changes----->+3|+/-4|-3|-2|-1|0

Melting Point
The elements in the middle of the periodic table have the highest melting point.
Noble Gases have the lowest melting point.
Starting from the <- and moving to the ->, the melting point increases until it gets to the middle of the table.

Atomic Radius
The radius decreases when moving on the table, up and to the right.
Francium has the largest atomic radius.
Helium has the smallest atomic radius.

Ionization Energy
Ionization Energy is the energy needed to completely remove an electron from an atom.
Ionization Energy increases going up and the right on the table.
All the noble gases have a high ionization energy.
Francium has the lowest Ionization Energy.
Helium has the highest Ionization Energy.
It's the opposite trend from atomic radius.

Electronegativity
Electronegativity refers to how much atoms want to electrons.
It has the same trend as Ionization Energy.

Density
A measure of the quantity of some physical property (usually mass)per unit length, area, or volume (usually volume).
Energy density is a measure of the amount of energy (often inthe form of electromagnetic radiation) per unit volume in a region of space or some material.
Thursday, October 28, 2010
Isotopes And Atoms
- Atomic Number - Number of Protons
- Mass minus Atomic number equals Number of Neutrons
- Isotopes - Some atomic number but different mass
- Mass Spectrometers -Are used to determine the relative abundance and mass of the isotopes of elements
Monday, October 25, 2010
Quantum Mechanics.
On friday, we learned about Quantum Mechanics!
The Quantum theory is that electrons are like a cloud of negative energy or a wave.
Orbitals are areas in 3d space where electrons are most likely there.
The energy of the electron is in its vibrational modes like notes on a guitar or a slinky!
Photons are produced when high energy modes change to lower energy modes.
S orbitals
each orbital holds 2 electrons.
P orbitals
There are 3 suborbitals
each contrain 2 electrons
total electrons is 6
D orbitals
there are 5 suborbitals
each contain 2 electrons
total electrons is 10
F orbitals
there are 7 suborbitals
each contain 2 electrons
total electrons is 14
The Quantum theory is that electrons are like a cloud of negative energy or a wave.
Orbitals are areas in 3d space where electrons are most likely there.
The energy of the electron is in its vibrational modes like notes on a guitar or a slinky!
Photons are produced when high energy modes change to lower energy modes.
S orbitals
each orbital holds 2 electrons.
P orbitals
There are 3 suborbitals
each contrain 2 electrons
total electrons is 6
D orbitals
there are 5 suborbitals
each contain 2 electrons
total electrons is 10
F orbitals
there are 7 suborbitals
each contain 2 electrons
total electrons is 14
Wednesday, October 20, 2010
Bohr's Diagram
The Bohr Model
Atoms are electrically neutral
There are 2 different models that can be used to represent the electron configuration of an atom:
1st: Energy Level Model
Example: 10 0
Ne
Neon
20 This would be represented like this:
8e
2e
20Ne 20= atomic mass
10 10= # of protons
2nd: Bohr Model
Example: 10 0
Ne
Neon
20 This would be represented like this:

Electrons occupy shells which are divided into orbitals
-2e in the first shell
-8e in the second shell
-8e in the third shell
-16e in the fourth shell
- etc.
Atoms are electrically neutral
There are 2 different models that can be used to represent the electron configuration of an atom:
1st: Energy Level Model
Example: 10 0
Ne
Neon
20 This would be represented like this:
8e
2e
20Ne 20= atomic mass
10 10= # of protons
2nd: Bohr Model
Example: 10 0
Ne
Neon
20 This would be represented like this:

Electrons occupy shells which are divided into orbitals
-2e in the first shell
-8e in the second shell
-8e in the third shell
-16e in the fourth shell
- etc.
Monday, October 18, 2010
Bohr's model
On October 15th, we learned about a man named Niel's bohr and how he came to create the Bohr Model.
Niels bohr wasn't satisfied with the model Earnest Rutherford created.His model was unstable and that protons and electrons should attract each other. So Bohr created a model based on the energy emitted by different atoms and that each atom has a specific spectra of light. To explain what he was talking about, Bohr stated that electrons occupy shells or orbitals.
Bohr's theory.
Electrons exist in orbitals
When they absorb energy, they move to a higher orbital to a lower one. They also release energy as a photon of light.
Here is a link to a program that shows how electrons move from shell to shell.
http://phet.colorado.edu/en/simulation/hydrogen-atom
Just click on it, click prediction and click on bohr. Why stop there? There are many other cool examples like
J.J thompsons plum pudding model and John Dalton's billard ball model!
-Julius Francia :)
Niels bohr wasn't satisfied with the model Earnest Rutherford created.His model was unstable and that protons and electrons should attract each other. So Bohr created a model based on the energy emitted by different atoms and that each atom has a specific spectra of light. To explain what he was talking about, Bohr stated that electrons occupy shells or orbitals.
Bohr's theory.
Electrons exist in orbitals
When they absorb energy, they move to a higher orbital to a lower one. They also release energy as a photon of light.
Here is a link to a program that shows how electrons move from shell to shell.
http://phet.colorado.edu/en/simulation/hydrogen-atom
Just click on it, click prediction and click on bohr. Why stop there? There are many other cool examples like
J.J thompsons plum pudding model and John Dalton's billard ball model!
-Julius Francia :)
Thursday, October 14, 2010
Atomic Theory
Democritus
- In 300BC Democritus said atoms were indivisible particles. It was not a testable theory, only a conceptual theory.
Lavoisier
- Law of conservation of mass
- Law of definite proportions
Proust
- If a compound is broken down into its constituents the procedure exist in the sam ration as the compound
Dalton
- Atoms are solid, indestructable spheres
JJ Thompson
- Rasin Bun Model
- Solid positive spheres, with negative particles embedded in them.
- First atomic theory to have positve protons and negative charges
Rutherford
- Show that atoms have a positive, with electrons outside it
- In 300BC Democritus said atoms were indivisible particles. It was not a testable theory, only a conceptual theory.
Lavoisier
- Law of conservation of mass
- Law of definite proportions
Proust
- If a compound is broken down into its constituents the procedure exist in the sam ration as the compound
Dalton
- Atoms are solid, indestructable spheres
JJ Thompson
- Rasin Bun Model
- Solid positive spheres, with negative particles embedded in them.
- First atomic theory to have positve protons and negative charges
Rutherford
- Show that atoms have a positive, with electrons outside it
Tuesday, October 12, 2010
Our First Lab :0
In our first lab, we conducted an experiment to see how much salt can be dissolved in various amounts of water then make a conclusion on how much salt could be dissolved in 200mL of water, using a graph. In this graph made a line of best fit and scrolled to 200mL and from there we took our estimate which was around 38.??g of salt, but our answer was not even close. The real answer was around 70.??g of salt. Though not so dangerous, in this lab we followed procedure and wore all safety gear and followed safety precautions. In this lab it is also important to ask what temperature of water to use because your results will reflect on how much salt can be dissolved in water.


The graph sort of looked like this but this is just an example:



The graph sort of looked like this but this is just an example:

Sunday, October 3, 2010
Density & Graphing
On Thursday, September 30th we learned how to calculate the density of an object and a couple of things about graphing.
The formula for density is:
density is usually expressed in the form Kilograms/Liters, Kilograms/meters cubed or grams/centimeters cubed.
There are 5 things a graph can have
1: labeled axis
2: Appropriate scale
3: A title
4:data points
5: a line of best fit
There are 3 things you can do with a graph
1: Read the graph
2: Find the slope
3: find the area of the graph.
Julius, Mark, Timothy
The formula for density is:
density is usually expressed in the form Kilograms/Liters, Kilograms/meters cubed or grams/centimeters cubed.
There are 5 things a graph can have
1: labeled axis
2: Appropriate scale
3: A title
4:data points
5: a line of best fit
There are 3 things you can do with a graph
1: Read the graph
2: Find the slope
3: find the area of the graph.
Julius, Mark, Timothy
Wednesday, September 29, 2010
Dimensional Analysis
Just like converting between curriences in Chemistry, it is usually necessary to convert between units
The process is called Dimensional Anaylis
Steps:
Find a unit equality
Find the converstion factors
Apply conversion factor
Cancel units
Taught Throughly Click the Link:
Scientific Notation & Significant Digits
Scientific Notation
Scientific Notation is used to show really big or really small numbers easily by an equation.
Example: 2000 = 2.0 x 10^3
4500000 = 4.5 x 10^7
0.000010 = 1.0 x 10^-5
Significant Digits
Significant Digits are used to be more precise and accurate with calculations.
-Precision and accuracy are very important
-Calculators are not smart enough to give a precise and accurate number
-Scientist have made rules to round off extra digits: These are a must!
-Non-zero digits are always significant
-If the zero is in the area to the right of the decimal then it is generally not significant
-0.0056
-Any digits to the left of a decimal point are significant
-5.00, 26.68, 18.1400
-Zeros after another number are significant
-2.00, 0.0002100, 5.870
Quick Note:
When Subtracting or Adding, round to the least precise number.
9.65471-6.21 = 3.44471
= 3.44
6.4798+3.254 = 9.7338
= 9.734
Si System, Percent Error and Experimental Accuracy
On monday, september 21st we learned about the Si System, Percent error and experimental accuracy.
When you use prefixes with Si Units,you can put a prefix in front of the unit and change the power of it.

The Si system uses many prefixes to represent very large or small numbers.
Note: Don’t mix up scientific notation and prefixes together, theres a big chance you will get confused.
Experiment Accuracy:
the maximum accuracy of any measurement is ½ of the smallest division of the measuring device.For example:A ruler with measurements of millimeters has a mamimum accuracy of + or – 0.5m.
Liquid in a gratuated cylinder will typically form a curved top called a MENISCUS. The volume is taken at the bottom of the meniscus.

Expressing error:
There are 3 types of error
- Physical error in the measuring device
- Sloppy measuring
- Changing ambient conditions
Usually you can’t prevent physical error and changing ambient conditions but you can prevent sloppy measuring.
Absolute error and percentage error:
To get absolute error, it is measured value – accepted value.
To get percent error:

Subscribe to:
Comments (Atom)










